BioChemistry > Research Paper > Research Paper > Exercise 1: BIOCHEMICAL CALCULATIONS (All)
Research Paper > Exercise 1: BIOCHEMICAL CALCULATIONS
Document Content and Description Below
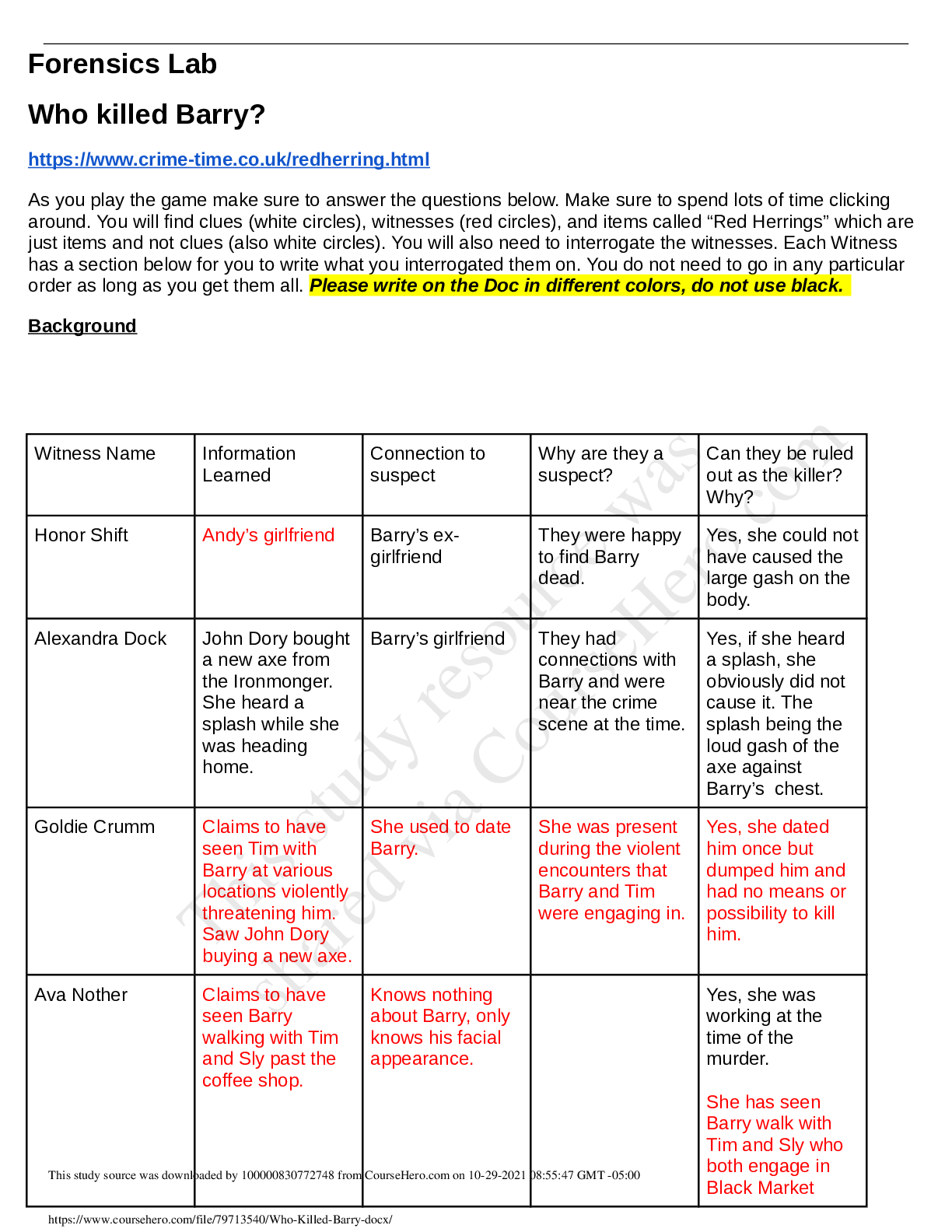
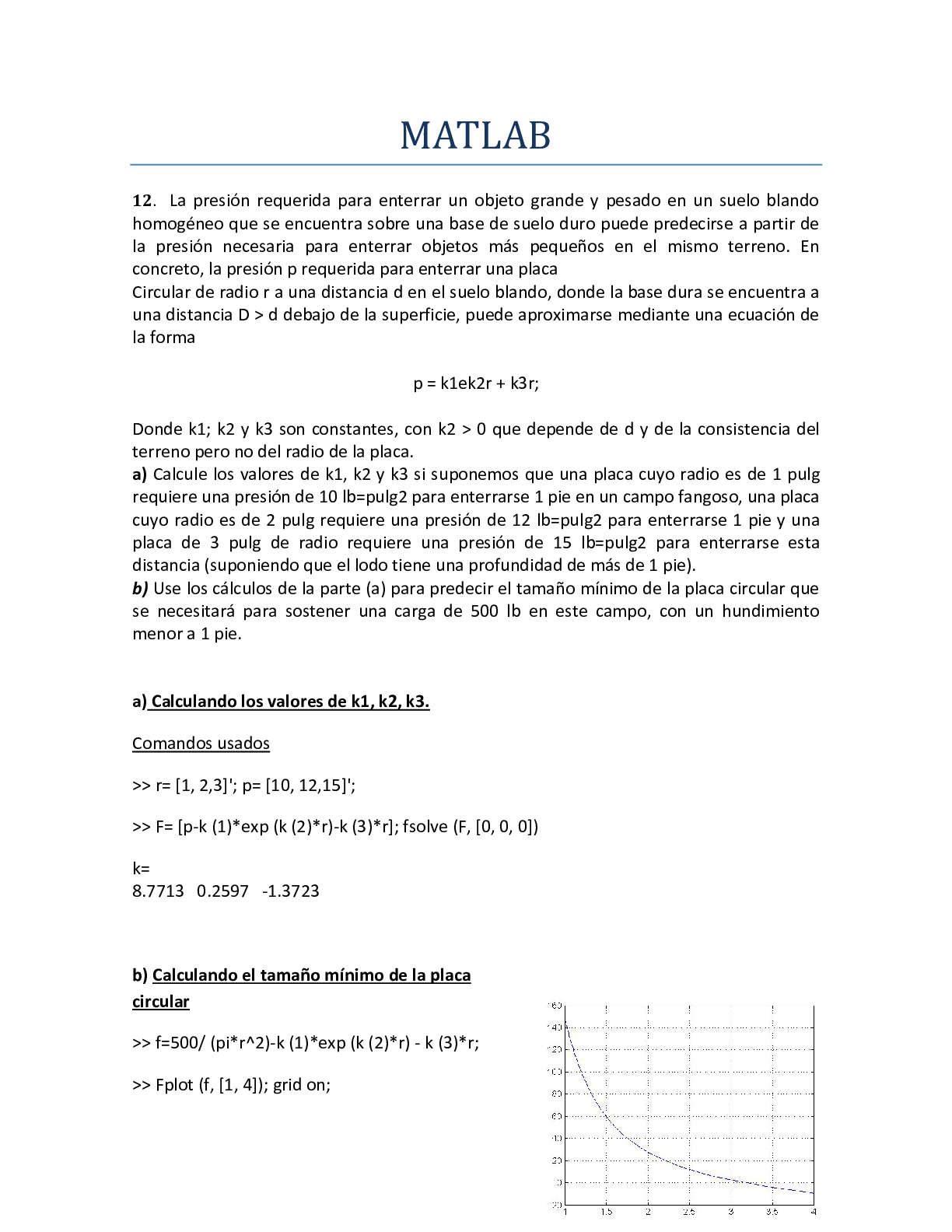
20EXERCISE 1 Arceño, Atis, Balo, Gonzales, Juntila, Manilag Purpose • To review on manipulation of numbers ... in chemical calculations • To review on conversion factors • To review on stoichiometric calculations 1. How many to. mmol in 720 mol 720 mol x 1000 mmol 1 mol = 720,000 mmol b. mg in 0.5 kg 0.5 kg | x 1000 g 1 kg x 1000 mg 1 g = 500,000 mg c. µL in 0.1 mL 0.1 mL | x 1000µL 1 mL = 100 µL d. dg in 70 g 70 gx 10 dg 1 g = 700 dg 2. Explain the difference between Mole is amount of chemical substance that contains as many representative particles while The molar mass is the mass of a given chemical element or chemical compound (g) divided by the amount of substance (mol) Mole and molar mass Molarity and Normality Molarity is a unit of concentration measuring the number of moles of a solute per liter of solution while Normality is a measure of concentration equal to the gram equivalent weight per liter of solution ppb and µg / L mass of a solute per 1000 million units of total mass, while the unit µg / L is usually used when calculating for% (w / v) 3. What is the equivalent of to. 6.2 µg in ppm 6.2µm x 1 mg | x 1 g g | 1000 µg 1000 kga. 7 ppb in µg L = 6.26 x 10-6 ppm = 7 µg L = 6.2 x 104 ppm in% (w / v) = 6.2 x 104mg x | 1 g | x | 1 L 1000 mL | x 100 L 1000 mg = 2.6% (w / v) 4. What weight in grams of caffeine (C8H10O2 N4) is required to prepare 10 mL each of the following solution? to. 7% (w / v) 7% (w / v) = ________g x 100 100 mL 7 g mL x 100 mL x __1__ 100 = 7 g b. 0.1 M 0.1 M = _____mol 0.1 L 0.1 mol L x 0.1 L x 196 g C8H10O2 N4 1 mol C8H10O2 N4 C = 12 x 8 = 96 H = 10 x 1 = 10 O = 2 x 16 = 32 N = 14 x 2 = 56 = 1.94 g 194 c. 2 mM 2 mM x __100 M_ 1000 mM x 0.1 L x 194 g C8H10O2 N4 mol = 0.0388 g d. 100 ppm 100 mg = ________mg L 0.1 L 1000 mg L x 0.1 L x 1.00 g 1000 mg = 0.01 g 5. Express the concentration in% (w / v) that is equivalent to 5.0 M NaOH solution. 0.5 mol NaOH L x 40 g NaOH 1 mol x 1.00 L 1000mL x 100 = 2% (w / v) NaOH 6. What weight of Ca (OH) 2 is necessary to prepare 3,500 mL of 0.7 M Ca (OH) 2 solution? [Show More]
Last updated: 1 year ago
Preview 1 out of 20 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Dec 12, 2020
Number of pages
20
Written in
Additional information
This document has been written for:
Uploaded
Dec 12, 2020
Downloads
0
Views
53




















Interdisciplinary Paper.png)










dfdfefe.png)

