Chemistry > QUESTIONS & ANSWERS > Student Exploration: Calorimetry Lab Vocabulary: calorie, calorimeter, joule, specific heat capacity (All)
Student Exploration: Calorimetry Lab Vocabulary: calorie, calorimeter, joule, specific heat capacity.
Document Content and Description Below
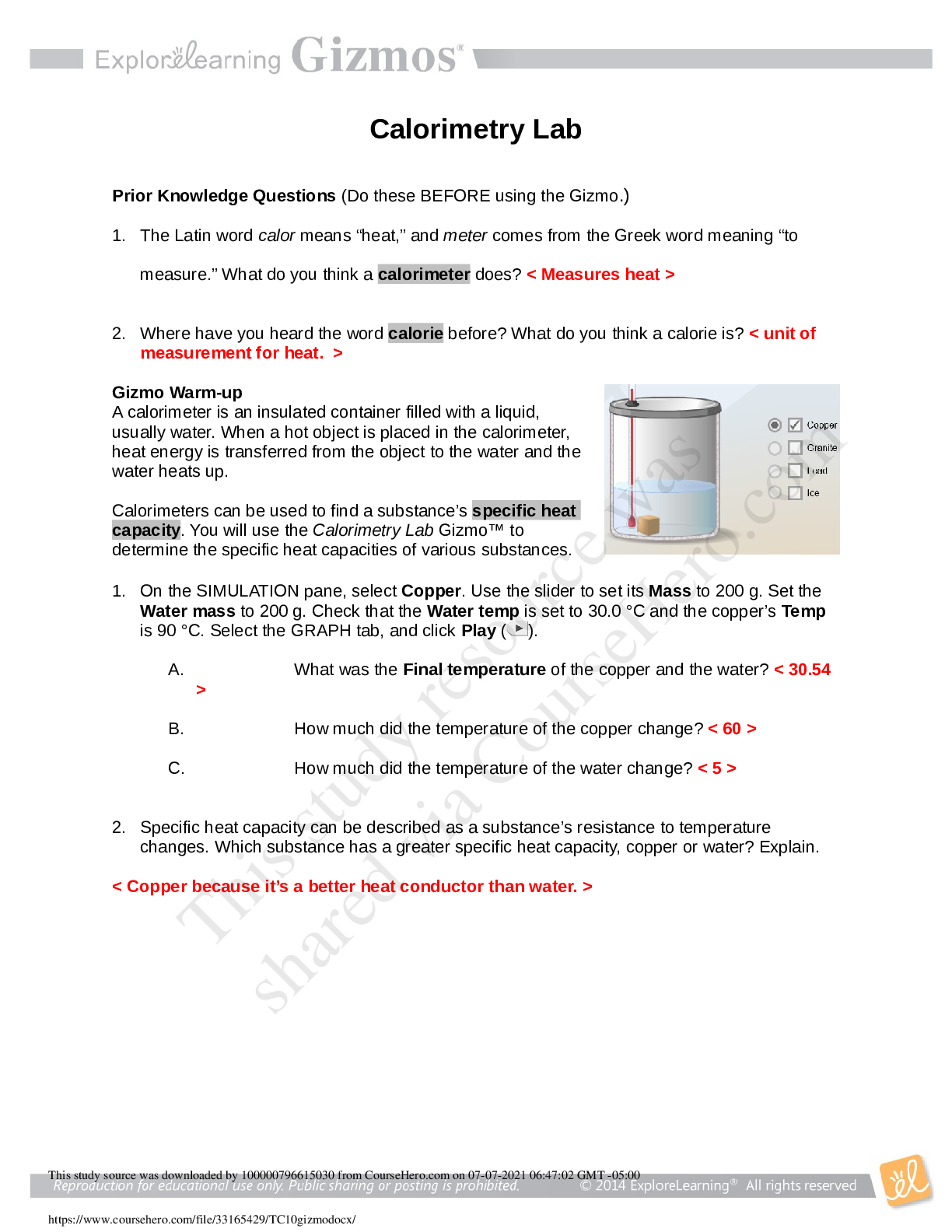
1. The Latin word calor means “heat,” and meter comes from the Greek word meaning “to measure.” What do you think a calorimeter does? < Measures heat > 2. Where have you heard the word calo... rie before? What do you think a calorie is? < unit of measurement for heat. > 3. On the SIMULATION pane, select Copper. Use the slider to set its Mass to 200 g. Set the Water mass to 200 g. Check that the Water temp is set to 30.0 °C and the copper’s Temp is 90 °C. Select the GRAPH tab, and click Play ( ). 4. Specific heat capacity can be described as a substance’s resistance to temperature changes. Which substance has a greater specific heat capacity, copper or water? Explain. 5. Predict: In the Gizmo warm-up, you saw how 200 g of 90 °C copper transfers heat to 200 g of 30.0 °C water. 6. Collect data: Use the Gizmo to determine the final temperature for each set-up listed below. Record your results in the tables. In the first table, you experiment with changing the water’s mass. In the second table, you change the copper’s mass. In the third table, you change the initial temperature of the copper. The first row of each table has been completed for you. [Show More]
Last updated: 1 year ago
Preview 1 out of 9 pages
Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
May 11, 2021
Number of pages
9
Written in
Additional information
This document has been written for:
Uploaded
May 11, 2021
Downloads
0
Views
41