Chemistry > STUDY GUIDE > CHEM 286 Theory Exam Covered Topics: SPECTROSCOPY, Labeling NMR spectrum, TECHNIQUES, and THEORETICA (All)
CHEM 286 Theory Exam Covered Topics: SPECTROSCOPY, Labeling NMR spectrum, TECHNIQUES, and THEORETICAL YIELD CALCULATIONS
Document Content and Description Below
CHEM 286 Theory Exam Covered Topics: SPECTROSCOPY Labeling IR spectrum: • Draw cmpd structure on spectrum, ID all fxnal groups present, assign important bands in spectrum (str + bends) • If s... everal bands exist for same fxnal group, use bracket to specifically cover correct set of signals • MEMORIZE Vibration type Functional Group Wavelength Characteristic OH stretch Alcohol/Phenol 3200-3600 Broad/strong Acid (-COOH centered ~3000) 2500-3200 Broad/strong NH stretch 3200-3500 Broad/strong CH stretch Sp2 3000-3100 Medium Sp3 2800-3000 Strong Aldehyde 2700-2850 Medium C triple bond C Alkyne 2100-2260 Weak C triple bond N Cyano 2110-2260 Medium C=O All 1660-1750 Very strong C=C Alkene/aromatic 1500-1650 Weak to strong Aromatic 1400-1515 Variable CH bend CH2/CH3 1350-1475 Medium/multiple bands OH bend Phenols 1315-1390 Medium-strong Acids 870-950 Broad/strong NH bend Amines/amides (weaker and sharper than OH) 1500-1650 Broad/strong 660-910 Broad/medium C-O stretch “Conjugated” (double-single partial bond character 1160-1310 Medium-strong Alcohol/ether 1000-1200 Medium C-N stretch 1020-1340 Weak-medium CH oop Polarized 950-1150 Strong Bends Aromatic 675-900 Strong • STRONG ACTIVATORS: groups w/ accessible lone pair e- (N, O) • WEAK ACTIVATORS: alkyl groups (inductive only), alkene/other aromatics (minor resonance) • STRONG DEACTIVATORS: groups w/ (+) charge, NO2, CN • MODERATE DEACTIVATORS: groups w/ carbonyl functionality • One oxygen = aldehyde/ketone • Aldehyde – look for signal in NMR (9-10ppm, SHARP, singlet/doublet –depends on neighbors) • Ketone – wide, broad signal • STRONGER SIGNALS = determined by POLARITY; H-BONDING WIDENS SIGNAL • Amides (NH, one loop) shifted closer to R than amines (NH2, 2 loops) on IR • If OH clearly separated = alcohol, if closer = phenol • CHECKLIST EXAMPLE • 4000-1300: look at CH stretch bands around 3000 • Are any/all to R of 3000? = Alkyl groups • Are any/all to L of 3000? = C double bond C/aromatic grp in molecule • Look for carbonyl in 1760-1690. If there is… • Is OH also present? = Carboxylic acid grp • C-O? = Ester • Aldehydic C-H? = Aldehyde • N-H = Amide • None of the above = Ketone • Look for broad OH band in 3500-3200. If there is, = alcohol/phenol • Other structural features to check for: • C-O stretches? = Ether (or ester if carbonyl present too) • C double bond C stretch? = Alkene • C triple bond C band? = Alkyne • If absence of major fxnal grps in 4000-1300, other than CH stretches, cmpd’s probably strict hydrocarbon Labeling NMR spectrum: • Draw cmpd structure on spectrum, assign letter to each H/group of equivalent H w/i molecule • Make sure to include: 1. Proton ID (based on structure – can’t do until structure is found, draw if not given) 2. # of H’s (based on integral) • Divide all by lowest integration value, round to nearest 0.5 then multiply all to get whole #’s (match to chemical formula) • 3 H integral = 3 substituents • 4H integral = 2 substituents • 5 H integral = 1 substituent 3. Splitting pattern (singlet, doublet, triplet, quartet, quintet, sextet, multiplet…etc.) • 2 doublets = para • N+1 (= *splitting pattern*, solve for n) rule for finding neighboring hydrogens • Hydrogen Deficiency Index (HDI): • # of H = [2 x (# of C)] + 2 • Each multiple bond or ring reduces H count by 2 (“degree of unsaturation”) – can be calc. from molecular formula using: • (2C + 2 + N – H – X) / 2 • C = carbons, N = nitrogens, H = hydrogens, X = halogens • If HDI=4, consider benzene ring • 7-8 ppm = Aromatic (i.e. benzene), 2.0-2.5=proton next to carbonyl • De-shielding (EWG) closer to 8ppm (maybe above) than 7ppm higher in aromatic region (De-shielded = L) • Shielded (EDG) might shift belo2w 7ppm (Shielded = R) • Hydrogen bonding hydrogens don’t couple (OH, NH, FH) TECHNIQUES • Reflux • Standard Reflux • The reflux temp is near bp of solvent • Place reagents in round bottom flask large enough to hold both reagents + enough solvent to dissolve them w/o being much more than ½ full • Choose solvent that… • Dissolves reactants @ boiling temp • Doesn’t react w/ reagents • Boils @ temp high enough to cause rapid rxn rate • Dissolve reactants in solvent and add stirbar • Place condenser upright on flask, connect to water faucet and run water through (in bottom, out top) • Put suitable heat source under flask. Reflux time starts when solution boils and solvent in flask starts condensing + falling back into flask • When rxn time’s up, turn off heat, let setup cool, dismantle, collect + purify product • Dry Reflux • Fill drying tube with drying agent – CaCl2, surrounded by loose cotton on either side • Have round bottom flask flame dried, then quickly place drying tube back onto flask (air moisture then trapped by drying agent) • Allow it to cool down, assemble apparatus as quickly as possible to avoid moisture condensing back • Quickly add reagents/solvents to reaction flask and assemble system • Carry out reaction like standard reflux • Extraction (same operation as washing EXTRACT good material from impure matrix, WASH impurities from good material) • Solutions must be insoluble in each other (to form distinct layers when mixed) • Something like 10% NaOH means it’s 90% water, therefore density will be very close to water’s • Examples: • Strong organic acid • Extract into sat’d sodium bicarbonate – foaming + fizzing b/c WB turns SA into salt that dissolves into water bicarbonate solution – invert funnel, point stem up and toward back of hood and open stopcock to vent funnel • To recover acid, add conc’d HCl until solution is acidic on litmus • To wash out SA, throw bicarbonate away • Weakly acidic organic acid • Extract into 10% NaOH water sol’n – SB needed to rip H off WA + turn them into salts that go into NaOH water layer • To recover acid, add conc’d HCl until acidic on litmus • To wash out WA, throw NaOH water sol’n away • Organic base • Extract w/ 10% HCl water sol’n – SA turns base into salt that goes into water layer • To recover base, add ammonium OH to water soln until basic on litmus • To wash out organic base, wash as previously noted and throw out soln [Show More]
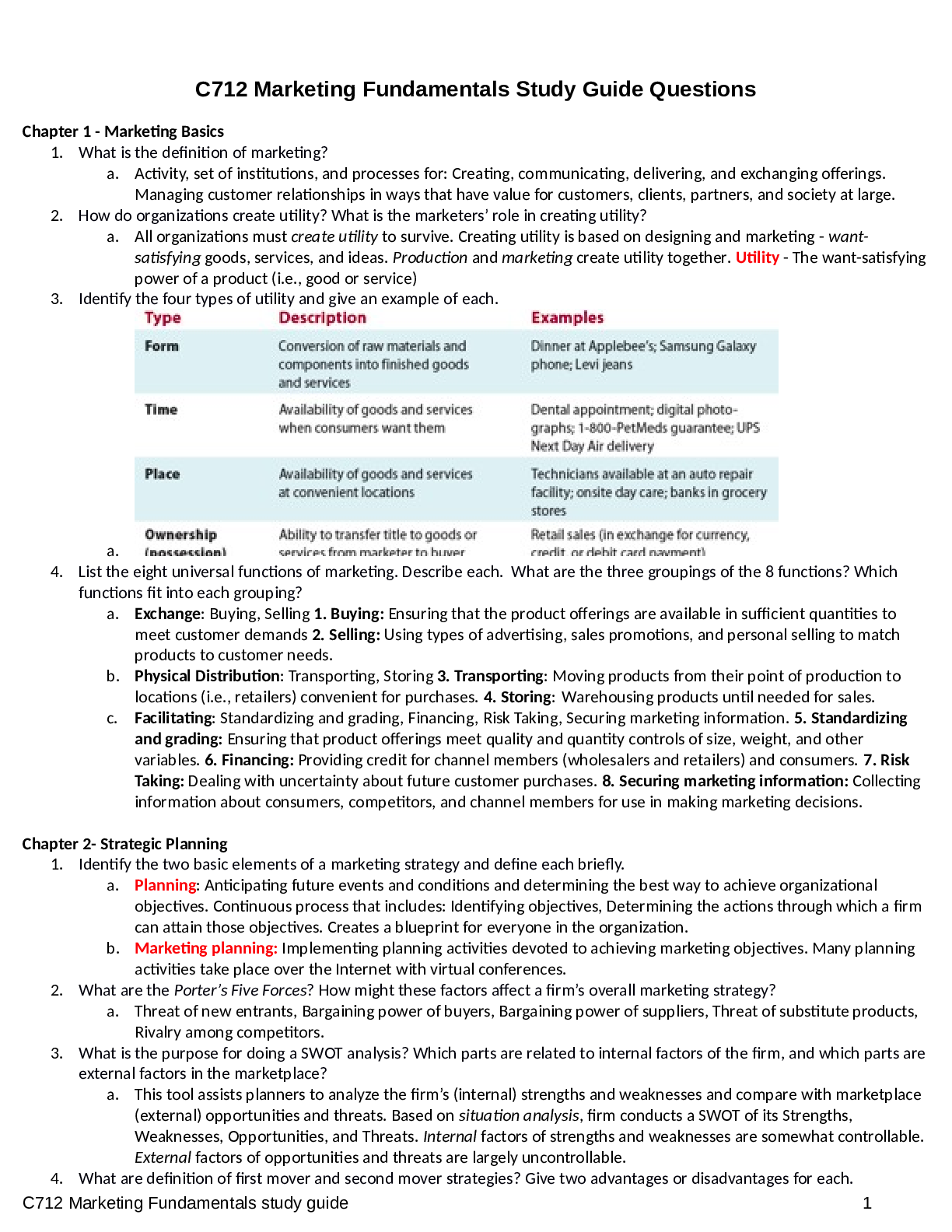
Last updated: 1 year ago
Preview 1 out of 5 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Apr 05, 2023
Number of pages
5
Written in
Additional information
This document has been written for:
Uploaded
Apr 05, 2023
Downloads
0
Views
49























.png)












