OCR > QUESTION PAPER (QP) > Ocr a level chemistry paper 3 2023 question paper (All)
Ocr a level chemistry paper 3 2023 question paper
Document Content and Description Below
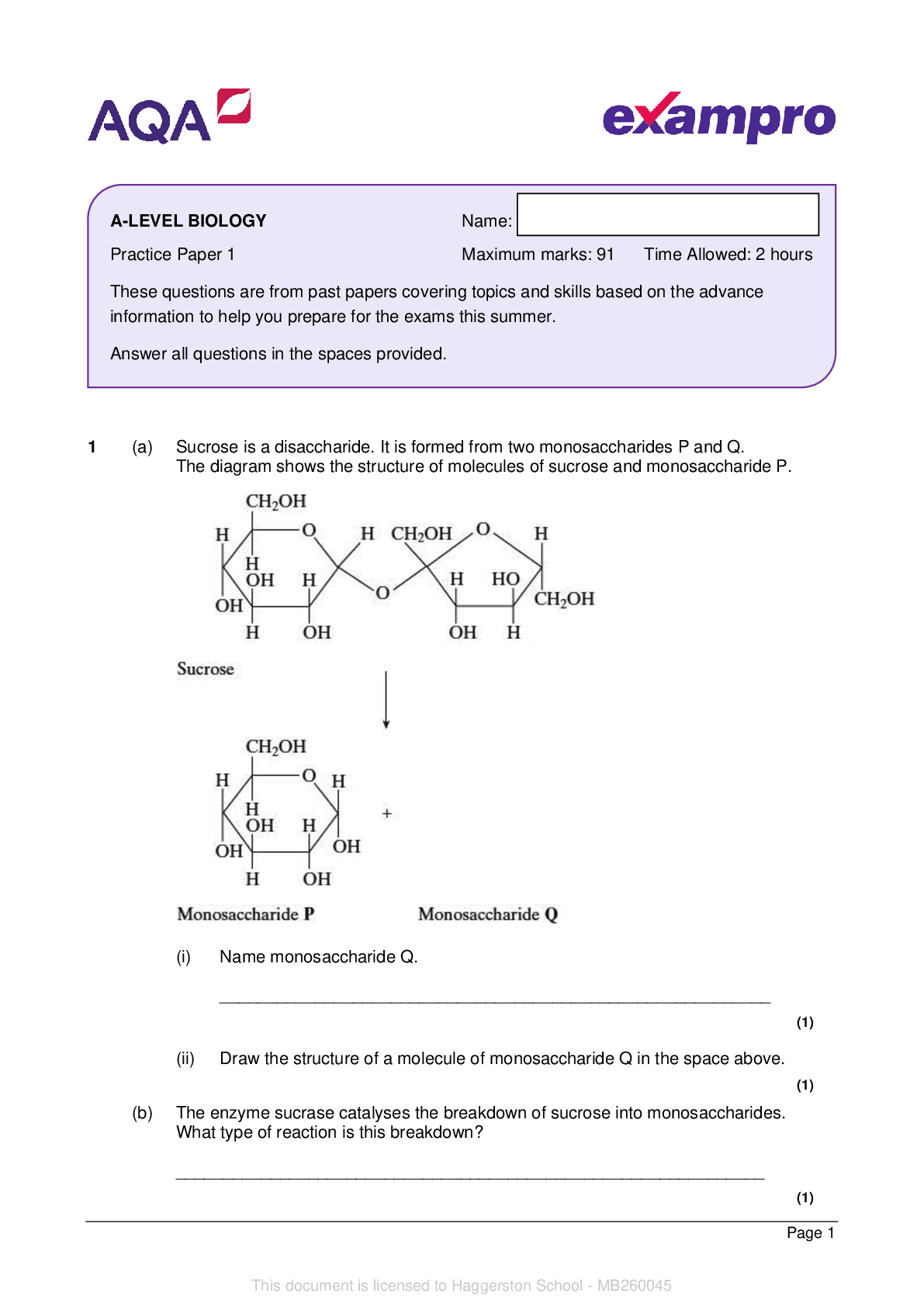
1 This question is about the first 36 elements in the periodic table: H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr From these 36 elements only... , write the symbol for the element(s) that fit each description. (a) The three elements that, when in a molecule, can form hydrogen bonds with other suitable molecules. .......................................................... [1] (b) The element in Period 2 with the successive ionisation energies shown below. Ionisation number 1st 2nd 3rd 4th 5th 6th 7th 8th Ionisation energy /kJmol–1 1000 2251 3361 4564 7012 8496 27107 31671 ................... [1] (c) An element that is a solid at RTP with a simple molecular lattice structure [Show More]
Last updated: 1 month ago
Preview 1 out of 24 pages

Reviews( 0 )
Document information
Connected school, study & course
About the document
Uploaded On
Mar 05, 2024
Number of pages
24
Written in
Additional information
This document has been written for:
Uploaded
Mar 05, 2024
Downloads
0
Views
26




.png)